Date : 06/10/2023
Relevance – GS Paper 3 – Science and Technology
Keywords – Nobel Prize, mRNA Vaccine, Indian Nobel Laureates, Ethical Concerns
Context –
The 2023 Nobel Prize for Physiology or Medicine has been conferred upon Katalin Karikó and Drew Weissman in recognition of their groundbreaking contributions to mRNA vaccine technology. The award highlights the immense societal impact of their work, although when solely evaluated from a scientific standpoint, the subset of mRNA vaccines employed during the COVID-19 pandemic might not entirely align with traditional criteria.
About Nobel Prize
Originating in Stockholm, Sweden, in 1901, the Nobel Prizes were established to honor the legacy of Alfred Nobel, a prominent Swedish inventor known for inventing dynamite and other powerful explosives.
Initially, the prizes were distributed in the fields of Physics, Chemistry, Physiology or Medicine, Literature, and Peace. In 1968, a sixth category was added, focusing on economic sciences, although it is not officially recognized as a Nobel Prize and is referred to as the Sveriges Riksbank Prize in Economic Sciences.
Nobel Prizes are typically awarded to individuals, with the exception of the Peace Prize, which can also be bestowed upon institutions. However, a maximum of three individuals can share a prize.
The Nobel Prizes are presented in Stockholm, Sweden, except for the Nobel Peace Prize, which is given in Oslo, Norway. Each prize includes a gold medal, a diploma with a citation, and a monetary award.
A significant milestone in the history of the Nobel Prizes was the achievement of Marie Curie, who became the first woman to win a Nobel Prize. Remarkably, she was honored twice with this prestigious award.
Indian Nobel Laureates
From 1913 to 2023, India has celebrated the accomplishments of nine Nobel Prize laureates. Rabindranath Tagore, recognized in 1913 for his deeply moving poetry, became India's inaugural recipient of this esteemed honor. Over the years, Indian excellence has been acknowledged across diverse fields:
- Literature: Rabindranath Tagore (1913)
- Physics: C. V. Raman (1930), Subrahmanyan Chandrasekhar (1983)
- Physiology or Medicine: Har Gobind Khorana (1968)
- Peace: Mother Teresa (1979), Kailash Satyarthi (2014)
- Economics: Amartya Sen (1998), Abhijit Banerjee (2019)
These laureates, spanning literature, physics, chemistry, medicine, peace, and economics, have left an indelible mark on the global stage. Their contributions not only bring immense pride to India but also serve as a beacon, inspiring future generations to strive for excellence in their chosen endeavors.
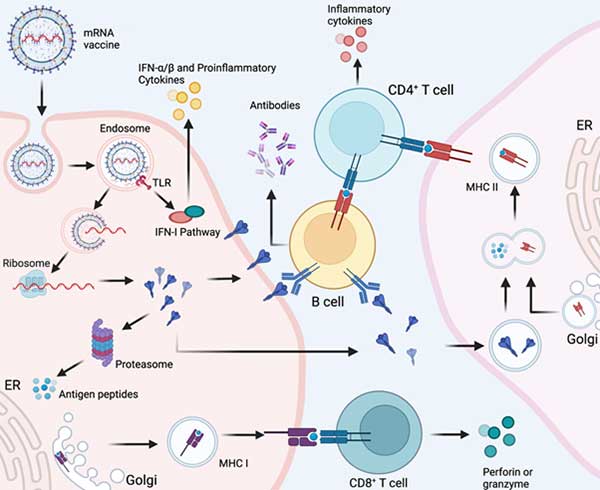
What is mRNA Vaccine?
mRNA vaccines operate by introducing a segment of messenger RNA (mRNA), a single-stranded RNA molecule essential in protein synthesis. This mRNA is synthesized from a DNA template during transcription.
These vaccines function by delivering a specific mRNA fragment corresponding to a viral protein, typically a small portion of a protein present on the virus's outer surface. Cells utilize this mRNA to generate the viral protein. As part of the body's natural immune response, the immune system identifies the protein as foreign and produces specialized proteins known as antibodies.
These antibodies play a crucial role in safeguarding the body against infections by recognizing individual viruses or other harmful microorganisms. They attach to these pathogens, labeling them for destruction. Notably, once antibodies are created, they persist in the body even after the pathogen has been eliminated. This persistence enables the immune system to swiftly respond if exposed to the same threat again.
Difference between mRNA and traditional vaccines
mRNA vaccines are a relatively new type of vaccine. They contain genetic material, or mRNA, that tells the body how to make a protein. This protein causes an immune response, which teaches the body how to protect itself from a specific virus. In contrast, traditional vaccines use weakened or dead microbes, or pieces of them, to stimulate immunity.
The Nobel Dilemma: mRNA Vaccines and Ethical Concerns
Scientific Collaboration and Commercialization
In the late 1990s, scientists Katalin Karikó and Dr. Weissman collaborated on mRNA research at the University of Pennsylvania. The University licensed its patents, leading to a cascade of sublicenses, eventually benefitting companies like Moderna and BioNTech, each paying substantial sums for access to this groundbreaking technology. Karikó's pivotal role continued, eventually contributing to the development of mRNA vaccines, notably in partnership with Pfizer for the COVID-19 vaccine.
Public Funding vs. Corporate Profits
A significant portion of the foundational research for drugs and vaccines relies on public funds. This essential stage involves risky and time-consuming exploration of potential biomolecular targets and suitable chemical candidates, often costing billions and spanning decades. Ironically, the same public whose taxes funded these discoveries often find themselves on the losing end when private companies capitalize on this research.
Global Access and Ethical Complexities
While companies like Moderna and Pfizer harnessed mRNA technology for COVID-19 vaccines, concerns arose over their prioritization of self-interest. They stockpiled vaccines for themselves before considering global distribution, creating challenges, especially for countries like India. Complications further emerged due to prolonged negotiations over pricing and liability issues, highlighting the ethical complexities of vaccine commercialization and global accessibility.
COVAX Initiative and Its Challenges
Overview of COVAX Program
The COVAX Program, standing for COVID-19 Vaccines Global Access, was a global endeavor orchestrated by the GAVI vaccine alliance. Part of the broader Access to COVID-19 Tools Accelerator initiated in 2020 by the WHO, the European Commission, and the government of France, COVAX aimed for fair and widespread access to COVID-19 vaccines. This effort was particularly focused on low-to-middle-income nations and was facilitated by UNICEF, leveraging its expertise as the world's largest vaccine buyer, managing vaccine procurement, logistics, and delivery.
COVAX's Struggles and Hindrances
Regrettably, the COVAX initiative faced significant setbacks in achieving its objectives. While India, Russia, and China exported substantial vaccine quantities, challenges emerged. India's manufacturing capacity was overestimated, causing production limitations. Quality concerns surfaced in Russia and China. Tragically, several African countries had to discard large quantities of vaccine doses due to their proximity to expiration dates, highlighting the complexities of global vaccine distribution.
The Corbevax Initiative: Dr. Karikó's Alternate Path
Discovery made by Dr Katalin Karikó and Drew Weissman
Karikó and Weissman noticed that dendritic cells recognized artificially produced mRNA as foreign, sparking inflammation. This raised questions about why this mRNA was deemed foreign while mRNA from mammalian cells didn't evoke a similar reaction. This observation prompted them to explore the unique properties distinguishing these two types of mRNA.
Innovative Vaccine Development
A team of researchers, including those from the Baylor College of Medicine in Houston, pioneered the creation of Corbevax, a protein sub-unit vaccine. Notably, this vaccine was unique because it was not patented, making it accessible for wider production and distribution.
Global Recognition and Ethical Acknowledgment
India's Biological E was granted the license to manufacture Corbevax. In a remarkable turn of events, the developers of this vaccine were nominated for the Nobel Prize for Peace in February 2022. Their nomination was based on their exceptional efforts in developing and disseminating an affordable COVID-19 vaccine without patent restrictions. This initiative received praise from Kenya’s Ambassador to the United Nations, recognizing the developers for their ethical and scientific leadership in addressing the global healthcare crisis.
Conclusion
Researchers should not be criticized for seeking financial gain from their efforts. Nevertheless, the mRNA vaccine narrative amid the COVID-19 crisis raised questions about their altruism. Despite the potential to benefit everyone during the pandemic, the technology's utilization fell short. Therefore, it is crucial for history to accurately document the events of the pandemic and contrast them with the narrative presented by the 2023 Nobel Prize in Medicine, highlighting any disparities.
Probable Questions for UPSC Mains Exam
- Discuss the Ethical and Global Accessibility Challenges in the Distribution of COVID-19 Vaccines, with Special Reference to the COVAX Initiative. (10 Marks, 150 Words)
- Examine the Impact of mRNA Vaccine Technology on Global Health Equity, Highlighting the Balance Between Scientific Advancements, Corporate Interests, and Public Welfare. (15 marks, 250 words)
Source – The Hindu